Goal
To develop a Microflow LC-Nanospray ESI-MS (MnESI-LC-MS) platform for targeted proteomics that achieves high throughput, high sensitivity, and robustness, by using Newomics® M3 Emitters and Thermo Fisher PepMap™ C18 columns on a Quantiva™ mass spectrometer for SRM analysis and a Q Exactive™ Plus mass spectrometer for PRM analysis.
Introduction
Liquid chromatography-mass spectrometry (LC-MS) is the enabling technology for the global-scale analysis of proteins (proteomics). In order to identify and quantify peptides and proteins in small volumes of biological samples, nanoflow LC-MS (flow rate < 1 mL/min) is routinely used in order to achieve high sensitivity. However, nanoflow LC-MS lacks robustness and throughput (i.e., speed). On the other hand, high-flow LC-MS (flow rate > 300 mL/min) typically achieves high throughput and robustness with a CV < 10% but lacks sensitivity. Microflow LC-MS (flow rate 1-50 mL/min) developed in the 1970s could potentially bridge this gap. In fact, there is a recent renaissance of microflow LC-MS because of the limited sample amounts for precision medicine applications. The Newomics award-winning silicon multinozzle emitters (M3 emitters) splits the incoming microflow evenly into multiple nanoflows, thereby dramatically enhancing the ionization efficiency to achieve high throughput, high sensitivity, and robustness for LC-MS. In this Application Note, we demonstrate the application of our microflow LC-nanospray ESI-MS platform (MnESI-LC-MS) for targeted proteomics studies of peptide mixtures spiked in human plasma digest matrix.
Methods
Sample Preparation
Human plasma digest sample was prepared using Thermo Scientific EasyPep™ Mini MS Sample Prep Kit (Cat.#: A40006). Dried peptides were stored in -80°C. During the experiments, the plasma digest sample was resuspended in Solvent A (3% acetonitrile in 97% water with 0.1% formic acid), and then the Thermo Scientific Pierce Peptide Retention Time Calibration (PRTC, Cat. #: 88320) mixture was added.
Data Processing
Raw data were imported into Skyline software (University of Washington) to obtain peak area intensities and retention times. Microsoft Excel was used to perform statistical analysis including relative standard deviation (RSD).
Instrumentation
The chromatographic separation was performed on the UltiMate 3000 nanoUPLC system (Thermo Fisher Scientific) with the following conditions (Table 1). The scheduled, selected reaction monitoring (SRM) analysis of peptides was performed on a TSQ Quantiva Triple Quadrupole Mass Spectrometer, and the scheduled parallel reaction monitoring (PRM) assay was achieved on a Thermo Scientific Q Exactive™ Plus MS. The position of M3 emitters relative to the ion transfer tube was tuned based on the TIC signal and was about 3~4 mm.
Results and Discussion
Sensitivity Improvement
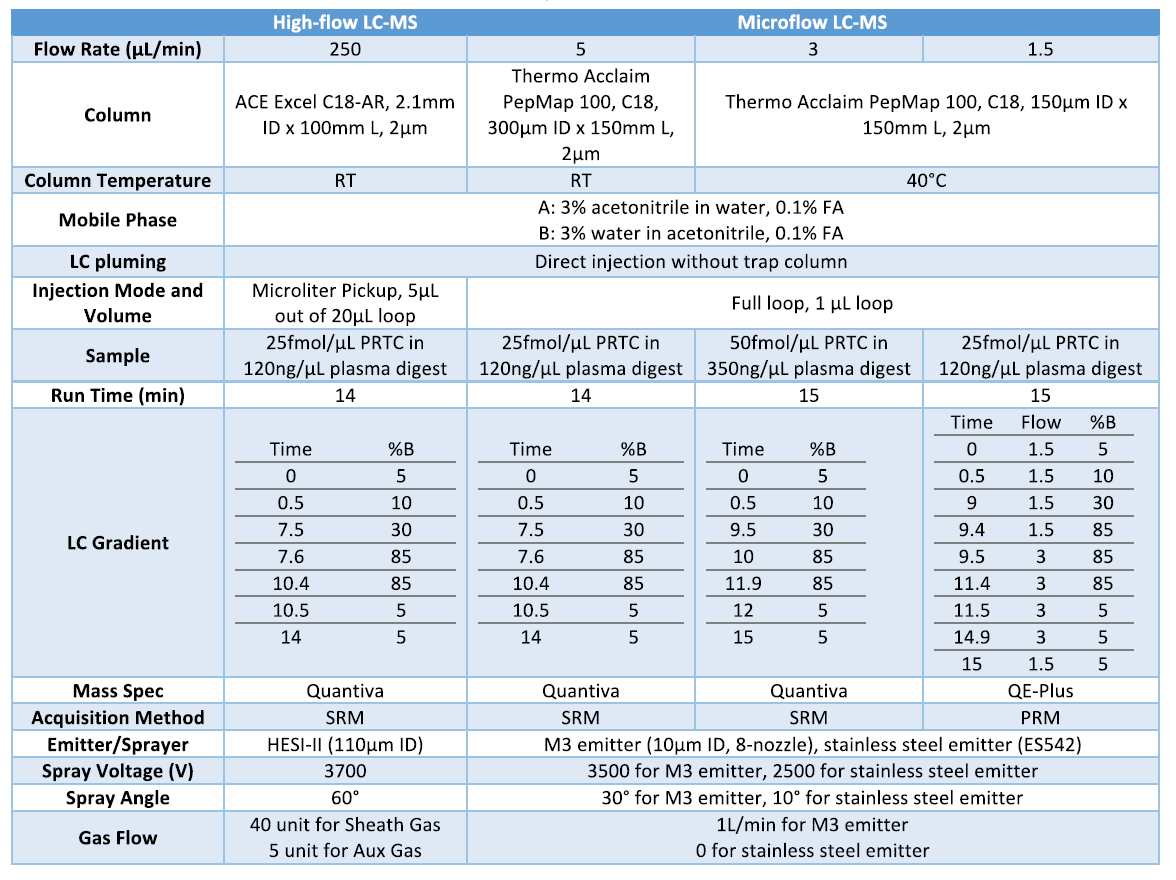
To demonstrate the performance of M3 emitters, we performed a head-to-head comparison between M3 emitters and stainless-steel emitters at microflow rates (5 µL/min) for targeted LC-MS analysis of PRTC peptides in plasma digest matrix. In addition, we compared the performance of microflow LC-MS by M3 emitters to high-flow LC-MS (2.1 mm column format). Figure 1 shows a representative extraction ion chromatogram of 15 PRTC peptides using M3 emitters at 5 µL/min. Excellent separation and detection of the pesticide compounds were achieved at microflow rates with the scheduled-SRM method. We determined the sensitivity improvement by comparing the peak area ratio for all 15 PRTC peptides, shown in Figure 2. Using the same column of 300 µm I.D. and the identical conditions, M3 emitters achieved the sensitivity gain of 2.3 on average over nanospray emitters (ES542). In addition, the average enhancement of micro-flow LC-MS by M3 emitter over high-flow LC-MS with HESI-II probe was about 48.9.
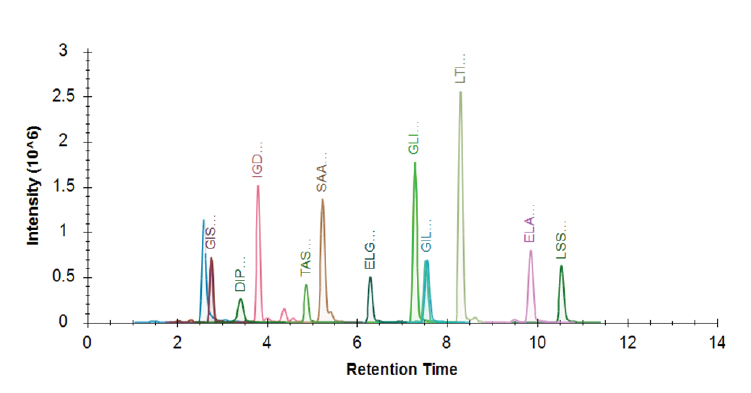
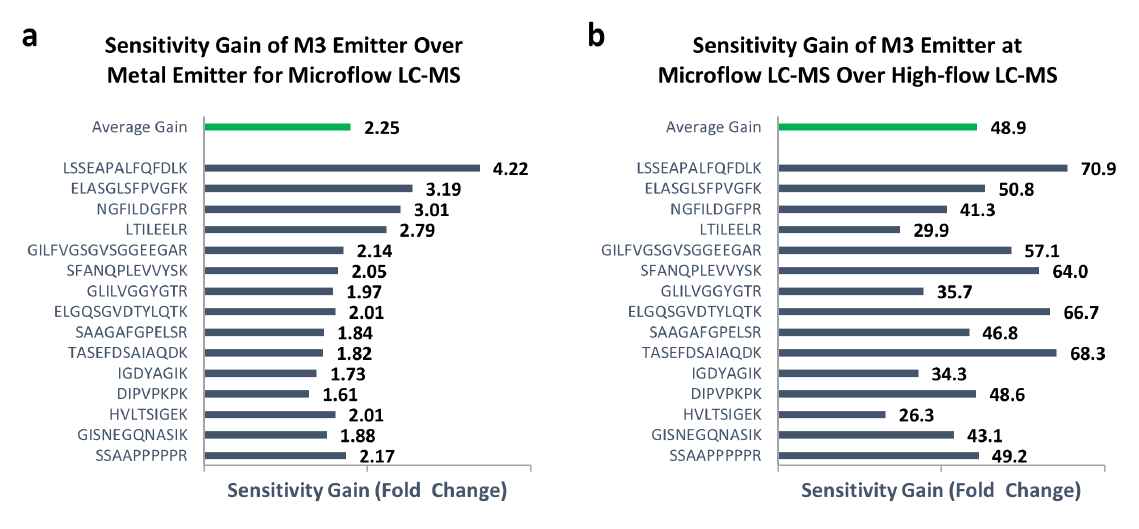
Robustness and Reproducibility
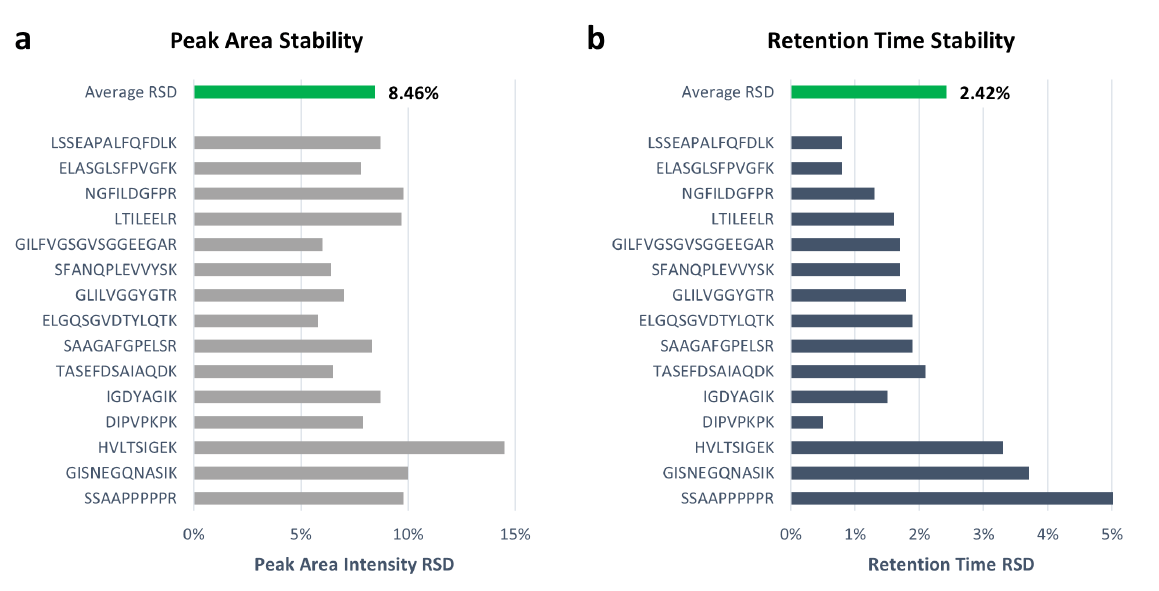
To determine the robustness of M3 emitters for microflow LC-MS analysis, we performed over 300 injections of plasma digests spiked with PRTC standards. Figure 3 shows the peak area stability and retention time stability for both micro-flow LC-MS and high-flow LC-MS analyses of PRTC peptides. For all 15 peptides, we achieved the average peak area intensity RSD of 5% for microflow LC-MS with M3 emitters even without using an internal standard to correct for MS signal variability. In addition, the peak area intensity RSD for the high-flow LC-MS was about 6% on average. The slightly better RSD from the M3 emitter might be due to the much stronger signals than high-flow HESI because of over 40-fold higher sensitivity. The average retention time RSD for M3 emitter was <1% over 300 injections, indicating excellent run-to-run repeatability, and was comparable to ~0.33% for high-flow LC-MS.
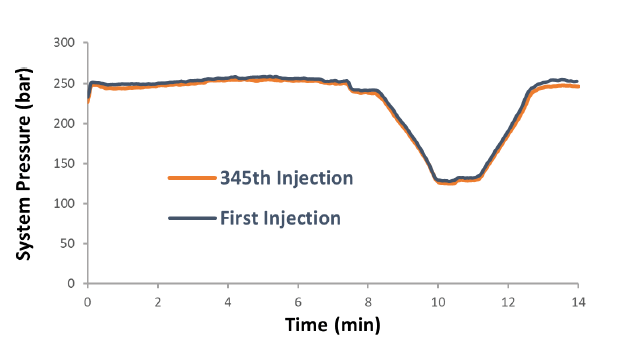
Very consistent back pressure was obtained after over 300 injections and we did not observe any pressure change from both the column and M3 emitter. Figure 4 shows the pressure trace from the first and last injection (#345). The M3 emitter still worked well after 345 injections and there was no sign of physical damage, clogging, or decreased performance. We have not carried out the emitter lifetime test yet but will investigate it in the future. These results demonstrated the rugged performance and excellent reproducibility of M3 emitters for microflow LC-MS, delivering the same level of high throughput and robustness as high-flow LC-MS.
Applications for High-Sensitivity and High-Throughput Proteomics
To achieve higher sensitivity for proteomics, it is straightforward to employ smaller diameter columns, such as 150 µm ID instead of 300 µm ID ones. In this study, we evaluated the robustness of microflow LC-MS at lower flow rates, by integrating M3 emitters with 150 µm ID columns. Figures 5 and 6 show peak area stability of 15 PRTC peptides for over 300 injections at flow rates of 3 µL/min and 1.5 µL/min, respectively. We achieved an excellent peak area precision of on average 5.2% RSD at 3 µL/min and 8.5% RSD at 1.5 µL/min, indicating the robustness of coupling M3 emitters with 150 µm ID columns for lower microflow LC-MS applications.
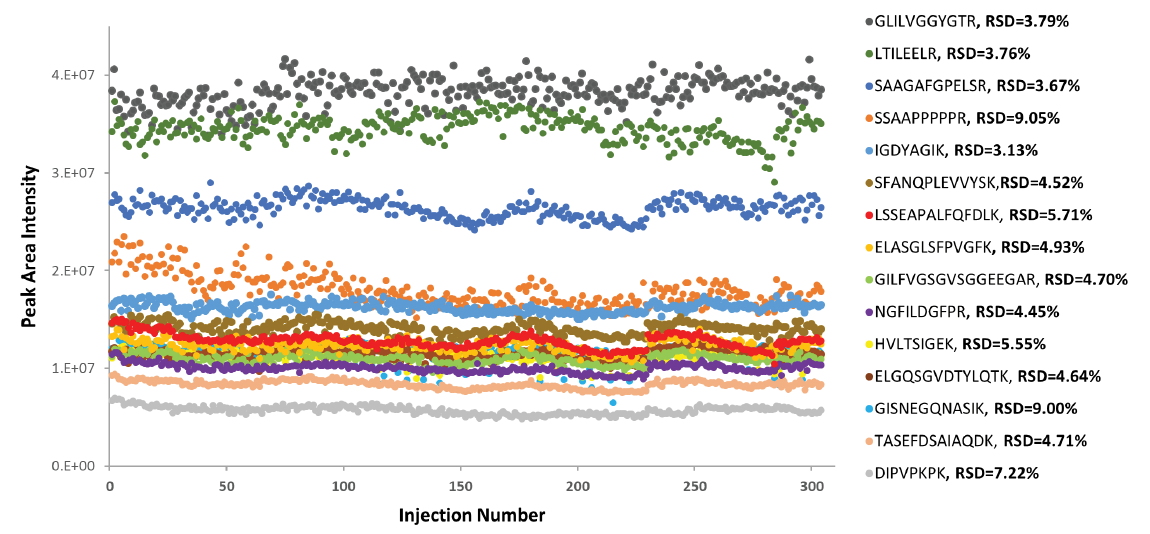
injections of 25 fmole PRTC peptides spiked in 120 ng human plasma digest was performed to obtain RSDs.
Conclusions
In summary, we have demonstrated that MnESI-LC-MS is a significant improvement over conventional microflow LC-MS. The robustness is on par with the gold standard of high-flow LC-MS and the sensitivity is approximately 50-fold higher than high-flow and 2.3-fold higher than conventional microflow LC-MS for proteomics. The combination of robustness and increased sensitivity has the potential to facilitate the wide adoption of microflow LC-MS for high-throughput applications in proteomics, metabolomics, lipidomics, and glycomics.
References
- Kim, Guo M., Yang P., Wang D., “Microfabricated Monolithic Multinozzle Emitters for Nanoelectrospray Mass Spectrometry,” (2007) Analytical Chemistry, 79(10), 3703-3707.
- Mao, Wang H.T., Yang P., Wang D., “Multinozzle Emitter Arrays for Nanoelectrospray Mass Spectrometry,” (2011) Analytical Chemistry, 83(15), 6282-6289.
- Mao, Gomez-Sjoberg R., Wang D., “Multinozzle Emitter Array Chips for Small-Volume Proteomics,” (2013) Analytical Chemistry, 85(2), 816-819.
- ASMS 2018 poster: po-65308-lc-ms-capillary-flow-sensitivity-proteomics-asms2018-po65308-en



